-
Notifications
You must be signed in to change notification settings - Fork 3
CARE SM Biobank
This module describes the data element in the Common Data Elemments group. These elements, defined by the JRC, can be found on the EU RD Platform at this link.
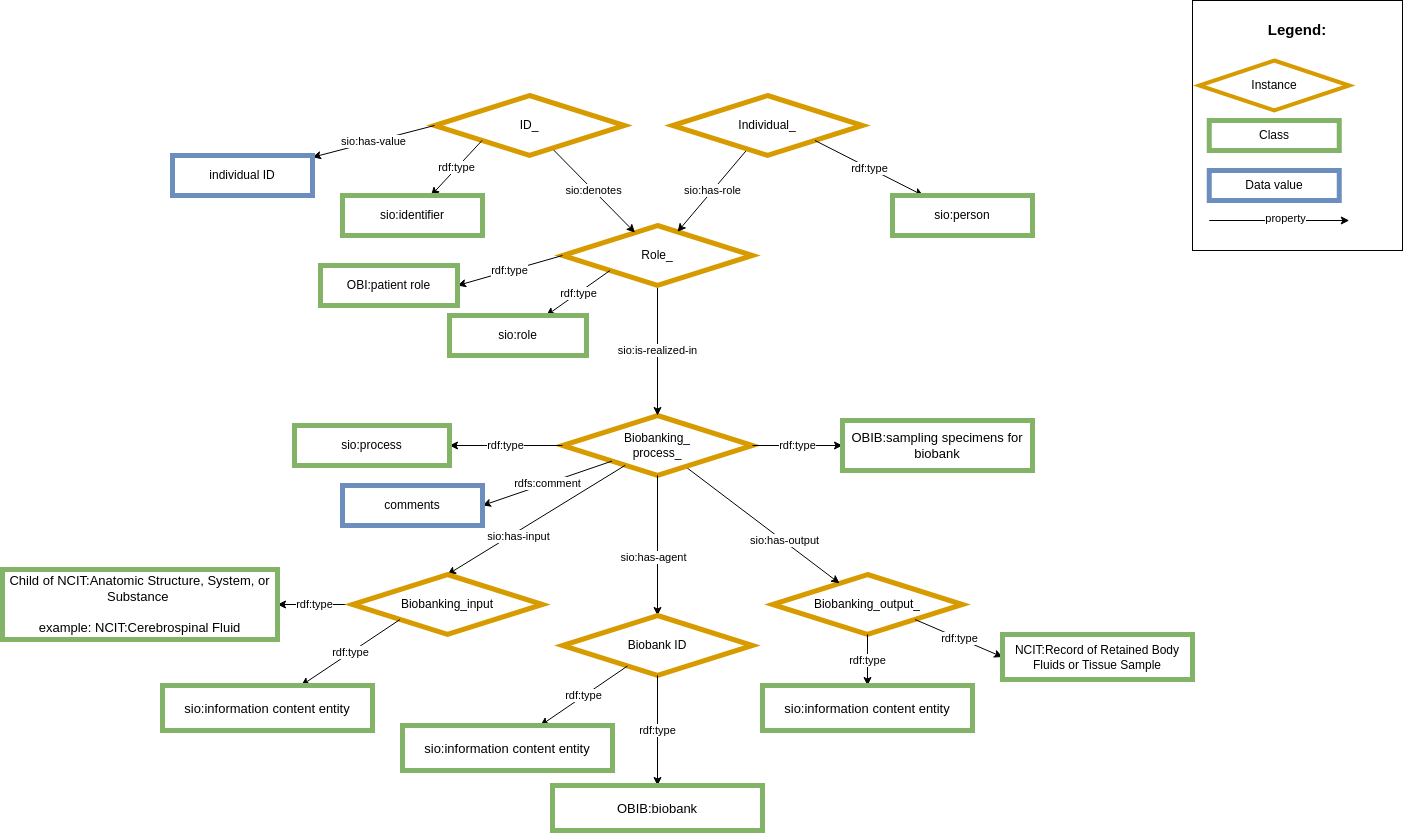
The module shows how the individual, defined as a patient, possesses a biological sample in a biobank, and its availability. This patient participates in a sampling procedure that has as output, the biological sample. In the module, a biobank participates as a target, measured as a global unique identifier.
CARE-SM is licensed under the MIT License - see the LICENSE file for more details.
-
List of defined data entries:
-
Medical history and participation status:
-
Demographic and questionnaire/PROMs representations:
-
Conditions and findings assesments:
-
Clinical and molecular measurements:
-
Treatment-related assesments:
-
Research sample availability and consent:
-
Clinical trials:
-